A nationwide collaborative research group, including Lecturer Mayuko Yamamoto Medical School, contributed to the approval of canakinumab for the treatment of Schnitzler syndrome.
Release Date:
Ilaris ® Solution for subcutaneous injection 150 mg Indications expanded to include Schnitzler's syndrome
― The findings that form the basis for this world-first expansion of indications were demonstrated in a physician-initiated clinical trial ―
京都大学大学院医学研究科・皮膚科学准教授・神戸直智、同・皮膚科学/先端医療基盤共同研究講座・特定准教授・中溝 聡、同・免疫・膠原病内科学准教授・吉藤 元、同・血液内科学/検査部・細胞療法センター助教・城 友泰、同・小児科学講師・井澤和司、および和歌山県立医科大学・分子遺伝学教授・井上徳光、兵庫医科大学・皮膚科学主任教授・金澤伸雄らの研究グループは、京都大学医学附属病院の他、高知大学医学部附属病院(皮膚科講師・山本真有子)、地方独立行政法人総合病院国保旭中央病院(アレルギー・膠原病内科部長・加々美新一郎および糖尿病代謝内科部長・竹村浩至)、一般財団法人神奈川県警友会けいゆう病院(皮膚科部長・河原由恵)との多施設共同治験として、日本人のシュニッツラー症候群患者に対するカナキヌマブ(商品名:イラリス®)の有効性と安全性を検討する医師主導治験(jRCT2051220139)を実施しました。その結果として得られた、2025年4月および10月に日本アレルギー学会の英文学術誌「Allergology International」に掲載された有効性と安全性に関する成果に基づいて、ノバルティスファーマ株式会社(以下、ノバルティス)は2025年5月に適応追加の申請を行い、2026年2月19日にシュニッツラー症候群への効能追加の承認を取得しました。
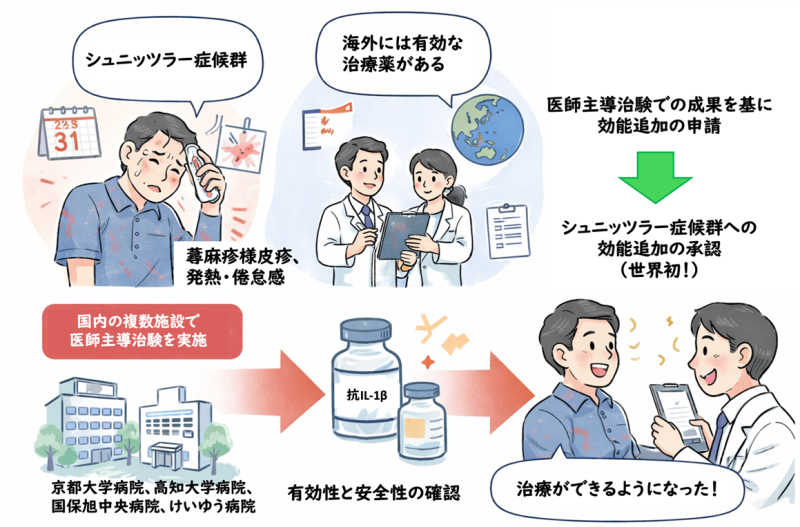
While effective treatments for Schnitzer syndrome—which causes urticaria-like rashes, fever, and fatigue—are available overseas, they were not previously available in Japan. Consequently, physician-initiated clinical trials were conducted at multiple domestic institutions, and based on the results, a new indication was added—the first of its kind in the world.(© 2026 Naotomo Kambe. Image created with ChatGPT5.2)
[Press Release] Indication Expansion for Ilaris® 150 mg Subcutaneous Injection for Schnitzler’s Syndrome
Title: Investigator-initiated, multicenter, single-arm, open-label study of the effectiveness of canakinumab in Japanese patients with Schnitzler syndrome.
(An investigator-initiated, multicenter, single-arm, open-label study evaluating the efficacy of canakinumab in Japanese patients with Schnitzler syndrome)
Authors: Kambe N, Yamamoto M, Takemura K, Kagami SI, Kawahara Y, Yoshifuji H, Jo T, Izawa K, Nakamizo S, Inoue N, Ito T, Amino Y, Ibi Y, Morita S, Kanazawa N.
Publishedin: Allergology International (English-language journal of the Japanese Society of Allergology) . 2025; 74(2): 254-262.
DOI: 10.1016/j.alit.2024.10.001.
Title: Neutrophils predominate as IL-1β-expressing cells in Schnitzler syndrome: Insights from the SCan study evaluating the efficacy and safety of canakinumab in Japanese patients.
(Neutrophils are the primary IL-1β-expressing cells in Schnitzler syndrome: Insights from the SCan study evaluating the efficacy and safety of canakinumab in Japanese patients)
Authors: Kambe N, Inoue N, Ueki Y, Zhou Y, Yonekura S, Katsuo K, Nakamizo S, Tsujimoto H, Ohtani K, Yoshifuji H, Jo T, Izawa K, Yamamoto M, Takemura K, Kagami SI, Kawahara Y, Amino Y, Ibi Y, Morita S, Kanazawa N.
Publishedin: Allergology International (English Journal of the Japanese Society of Allergology). 2025; 74(4): 605-615.
DOI: 10.1016/j.alit.2025.04.003.